Back to overview
Salt, Sodium Chloride, Potassium Chloride
For our daily bread
From earliest times on not only humans used rock salt to equalize the mineral balance of the body. For this purpose, also animals in the wilderness regularly visit salt deposits. Already rather early salt was artificially produced from salt springs or seawater. Salt was as valuable as gold. This can be derived from the fact that it became a rather precious commodity for which it was worth to accept the trouble and expenditures transporting it over long distances. The trade routes were called “Salt streets”. Both, the cities where salt was won and the owners of the salt production facilities obtained wealth and power. Today many city names still refer to the salt production: Salzburg, Bad Salzungen, Bad Reichenhall etc. Beginning during the middle of the 19th century, motivated through the work of Justus von Liebig, the mining production of potassium salt gained in significance, especially the production of mineral fertilizer.
Natural salt cavities
The result was the search for salt cavities. Today there are extensive salt cavities known in Germany and also worldwide. Generally natural salt cavities develop by evaporation of ocean and fresh water in hot and dry (arid) climatic conditions. The in the water dissolved minerals crystallize during evaporation of the water in dependence of their solubility and deposit in layers on the ground. In this manner developed, for example approximately 250 million years ago, the domestic salt cavities through evaporation of a flat sea, the Zechsteinmeer, at which there were several cycles of evaporation and the afflux of fresh seawater. This caused that already deposited salts were partially dissolved again respectively recrystallized. Through this and often due to later tectonic influences, additional salt minerals were created, which were not present in the primary deposits. Similar processes were and are still occurring worldwide today.
The complex processes of the formation of salt cavities result in that from a few main elements like sodium, potassium, magnesium, calcium, chloride, sulphate and water, numerous salt minerals can be created. The most important are:
| Mineral names | Chemical Composition |
| Halite, rock salt, common table salt | NaCl |
| Sylvine | KCl |
| Carnallite | KCl . MgCl2 . 6 H2O |
| Kieserite | MgSO4 . H2O |
| Polyhalite | K2SO4 . MgSO4 . 2 CaSO4 . 2 H2O |
| Kainite | 4 KCl . 4 MgSO4 . 11 H2O |
| Anhydrite | CaSO4 |
| Gypsum | CaSO4 . 2 H2O |
Almost 20 additional salt minerals like langbeinite, schönite, bischofite, leonite etc. can occur in the evaporates. They form the evaporates in the most different mineral compositions, from pure rock salt to the simultaneous combination of eight and more salt minerals.
Utilizing of the deposits
Rock salt and potassium find versatile applications as fertilizer, as the basic chemicals of many chemical processes, in the food industry, in the medical field, in spa applications and much more. Here it depends through specific retrieval and production processes to provide the desired products in the required quality. The variety of evaporates has effects on the selection of the suitable extraction methods, the mining safety and the required manufacturing processes. A prerequisite for an efficient salt extraction and processing is the exact knowledge of the mineral compositions of in-situ evaporates, as well as their chemical composition and also of the accruing intermediate and final products during the manufacturing process.
Analysis of evaporites
A very elegant method for the determination of the mineral composition is the X-ray powder diffraction.
The K-UTEC AG Salt Technologies located in Sonderhausen/Germany, which possess a more than 50 year treasure trove of experiences in research of salt mining and processing, conducts such analyses on a large scale. The mineral composition is determined with the assistance of an X-ray Powder Diffractometer “D2 Phaser from Bruker AXS” in Karlsruhe/Germany by analysing characteristic X-ray diagrams, as well as with the assistance microscopic procedures. The chemical analysis provides the element content from which the quantities of the minerals contained can be calculated.
Production of the measuring sample
The salt cavities are due to the conditions during formation mostly very inhomogeneous. Therefore it is often necessary to analyse larger sample amounts, often several kg. Only then correct information about the average mineral content of a certain area of a salt deposit of crude salts or intermediate or end products can be obtained.
Since for the chemical analysis less than 50 g and for the mineral determination even only 3 g of the entire sample are needed, a special emphasis is on the sample comminution and homogenization. As a first step, a pre-comminution with the Jaw Crusher PULVERISETTE 1 is recommended. The produced material is relatively inhomogeneous with a particle size of approximately 4 mm and must be mixed well. Then a representative sample of 200 to 500 g is to be drawn. For the exact division of the sample, the Rotary Cone Sample Divider LABORETTE 27 is recommended. A sub set serves as a reference sample and an additional sub set is further comminuted with the Variable Speed Rotor Mill PULVERISETTE 14 classic line using a 1 mm sieve. The produced material can immediately be subject to a chemical analysis.
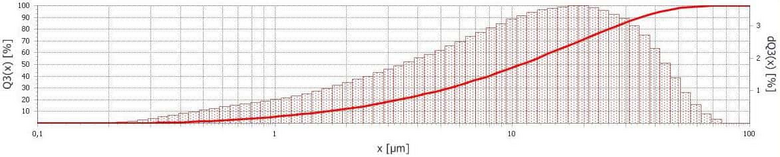
For the X-ray powder diffraction again a smaller sub set is comminuted with the Variable Speed Rotor Mill PULVERISETTE 14 using a 0.08 mm sieve. A loose and a very pourable powder is obtained which can be immediately placed on the sample carrier of the measuring instrument. The sample obtained in this manner, very well fulfils the recommendations in order to be measured with the X-ray Powder Diffractometer D2 PHASER. For the measurement the particle size is a decisive factor. Known from experience a particle size below 63 µm is to be assumed. In order to evaluate the quality of the processing steps, the particle size distribution was determined with the Laser Particle Sizer ANALYSETTE 22. In general, with the comminution of Variable Speed Rotor Mill, a distribution curve of 50 to 60 % smaller than half of mesh width of the sieve is expected. With very brittle materials, a clearly higher fineness is attained. Here for example:
Particle size distribution of the salt sample
- Comminuted with the Variable Speed Rotor Mill PULVERISETTE 14 classic line using a des 0.08 mm sieve
- Measured with the Laser Particle Sizer ANALYSETTE 22 MicroTec plus
This is a very impressive result, since the comminuted materials are only 50 to 60 % smaller than half a mesh width of the used sieve. It was tried to grind the salt with Planetary Ball Mills, this did not work so well. Salt quickly sticks to the walls of the grinding bowl, preventing a further comminution. Possibly the high application of energy in connection with the water of crystallisation of several mineral phases is the cause of this.
Benefit of the Method
The determination of the qualitative and quantitative mineral composition via the classic method is very elaborate. With the introduced method, in comparatively brief time it is possible to analyse the samples in regard to quality and in broad limits in regards to quantity. For small mineral contents below 1 % which can definitely be a decisive factor for the production, often only the classic evaluation with a microscope remains. The largest source of mistakes is the inhomogeneity of the cavities and unavoidable mistake during sampling. In this regard are the unavoidable mistakes during the sample preparation and the quantitative evaluation of the X-ray crystallography are relativized.
-
Download the FRITSCH-report as PDF file
-
Detailed grinding reports
Back to overview